Mass of electron: The electron mass (me) is the mass of a stationary electron, also known as the rest mass of the electron. The value of mass of an electron is, 9.109 383 7015 x 10-31 kg, 0.00054858 amu.
Mass of Electron
The electron mass determines a number of observed effects in atomic physics, so its very imporatant to find out the most accurate value of electron mass.
The mass of the electron was determined directly from combining two measurements.
- The mass-to-charge ratio of the electron
- The charge of the electron
The mass-to-charge ratio of the electron was first estimated by Arthur Schuster in 1890 .
in 1897, J. J. Thomson showed that cathode rays consist of streams of particles later called electrons, and made more precise measurements of their mass-to-charge ratio using a cathode ray tube.
The charge of the electron was determined by Robert A. Millikan in his oil drop experiment in 1909.
Mass of Electron,Proton and neutron is given. Another detailed post : Electron Mass – Mass of electron, Charge, Speed, & value of electron can be read here
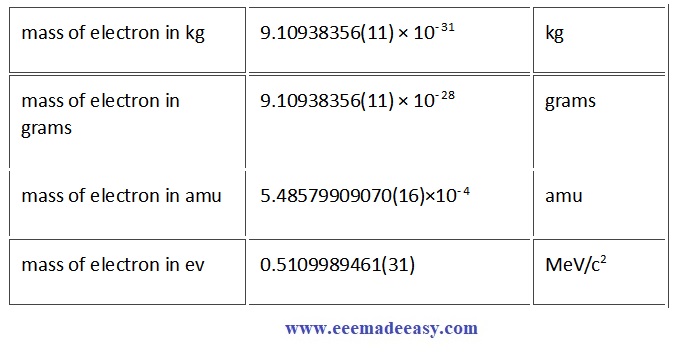
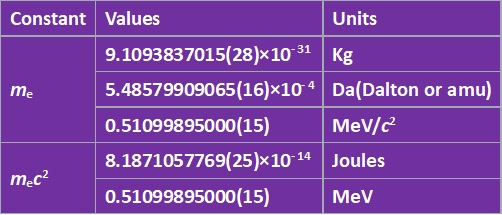
Electron Mass and uncertainty
| electron mass | me |
|---|---|
| Numerical value | 9.109 383 7015 x 10-31 kg |
| Standard uncertainty | 0.000 000 0028 x 10-31 kg |
| Relative standard uncertainty | 3.0 x 10-10 |
| Concise form | 9.109 383 7015(28) x 10-31 kg |
Mass of Proton
Proton mass mp = 1.672621898×10−27 Kg. Mass of proton is higher than the mass of electron.
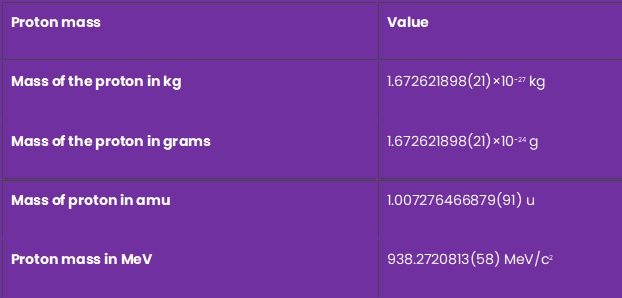
| Proton mass | Value |
|---|---|
| Mass of the proton in kg | 1.672621898(21)×10−27 kg |
| Mass of the proton in grams | 1.672621898(21)×10−24 g |
| Mass of proton in amu | 1.007276466879(91) u |
| Proton mass in MeV | 938.2720813(58) MeV/c2 |
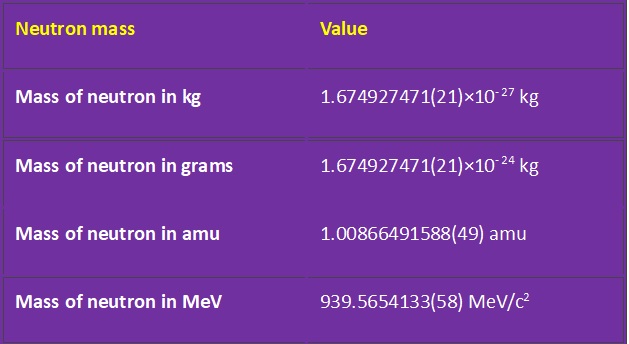
Mass of Neutron
Mass of Neutron is , mN 1.674927471 x 10-27kg.
| Neutron mass | Value |
|---|---|
| Mass of neutron in kg | 1.674927471(21)×10−27 kg |
| Mass of neutron in grams | 1.674927471(21)×10−24 kg |
| Mass of neutron in amu | 1.00866491588(49) amu |
| Mass of neutron in MeV | 939.5654133(58) MeV/c2 |
Comparison of Mass of Electron, Proton and Neutron

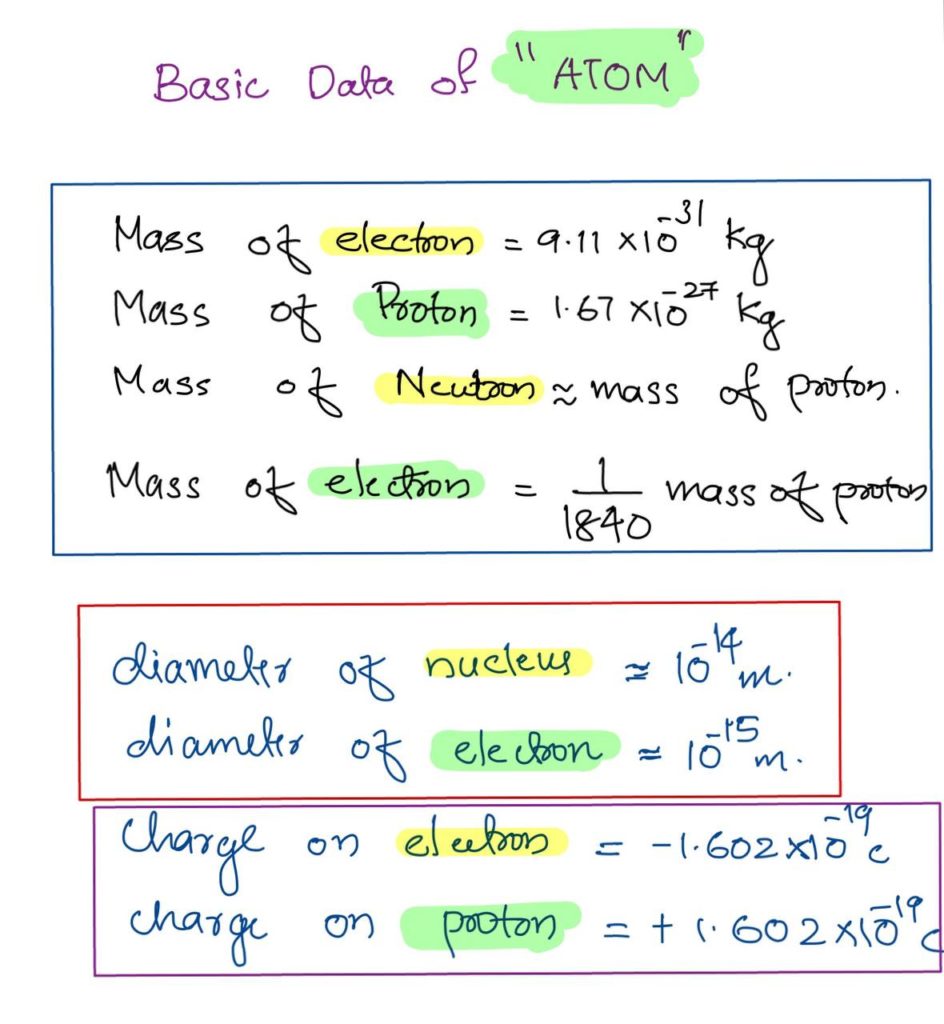
Basic Physical Concepts of Atom, Nucleus, Proton, electron
Electron-Mass of electron, Charge of electron, Speed of Electron, value of electron
Basic Physical Concepts of Atom, Nucleus, Proton, electron is detailed in this post in EEE Notebook Read it HERE
Download & Install EEE Made Easy App
Important FAQ’s
what is the mass of an electron?
What is mass of electron and proton?
How much is mass of electron in kg?
What is mass of electron in amu?
What is mass of electron in grams?
What is electron rest mass?
What is the value of the charge of an electron?
Latest Posts in EEE Made Easy
- Compensating WindingsCompensating Windings: Compensating Windings are used for large direct current machines which are subjected to large fluctuations in load i.e. … Read more
- Transformers Electrical Engineering Interview QuestionsTransformers Electrical Engineering Interview Questions: QUESTIONS AND ANSWERS ON TRANSFORMERS Read: 125 Electrical Interview Questions Q.1. How is magnetic leakage … Read more
- DC Motor Electrical Engineering Interview QuestionsDC Motor Electrical Engineering Interview Questions: DC Motor Interview Questions and answers. Read: 125 Electrical Interview Questions Q. 1. How … Read more
- Special Electrical Machines Interview QuestionsSpecial Electrical Machines Interview Questions: Electrical Interview Questions Read: Electrical Interview Q.1. Do stepper motors have internal or external fans? … Read more
- 125 Electrical Engineering Interview QuestionsElectrical Engineering Interview Questions: Read 125 Questions based on Electrical Interview Questions. 1) What is Electrical Engineering? Electrical Engineering … Read more
- Syllabus Training Instructor Plumber|14/2025 Syllabus Kerala PSCSyllabus Training Instructor Plumber: 14/2025 Syllabus Kerala PSC. Download the syllabus and Previous question papers for the Training Instructor in … Read more
- [PDF] Syllabus AE KSEB Transfer|378/2025 Syllabus Kerala PSCSyllabus AE KSEB Transfer: DETAILED SYLLABUS FOR THE POST OF ASSISTANT ENGINEER(ELECTRICAL) (Kerala State Electricity Board Ltd.) – By Transfer … Read more

